Introduction
Celiac disease is a systemic immune-mediated disorder of the intestine caused by ingesting foods containing gluten. Gluten consists of two essential proteins, namely Gliadin and Glutenin. Gliadin is a prolamin that plays a vital role in inducing celiac disease. Prolamins are plant proteins that are difficult to digest fully. Scientists have found that in patients with celiac disease, either the HLA-DQ2 or HLA-DQ8 genes trigger the CD4+ T-lymphocyte when exposed to epitopes of prolamine. In this article, we will be trying to understand the process of the development of celiac disease.
What are Prolamins?
As discussed above, prolamins are plant proteins that are difficult to digest fully. Prolamins make up most of the proteins in food and are responsible for immunological response in patients with celiac disease. Prolamins are proline (prol-) and glutamine (-amin)-rich storage proteins that dissolve in alcohols and are resistant to intestinal proteases and peptidases. Prolamins are found in cereal grains containing wheat (gliadin), barley (hordein), rye (secalin), and oats. All of these plant-based food items possess separate but related prolamins. The very high glutamine and proline content in the gliadins and glutenins of wheat and the hordeins and secalins play a crucial role in celiac disease pathogenesis.
Pathophysiology of celiac disease
The celiac disease appears to be multifactorial, in the sense that it can be caused by more than one hereditary component, or it may take more than one factor for the disease to develop in a person. The pathophysiology of celiac disease can be genetic, or environmental, or immunological. Here we have tried to explain the pathophysiology of celiac disease in a simple way as possible.
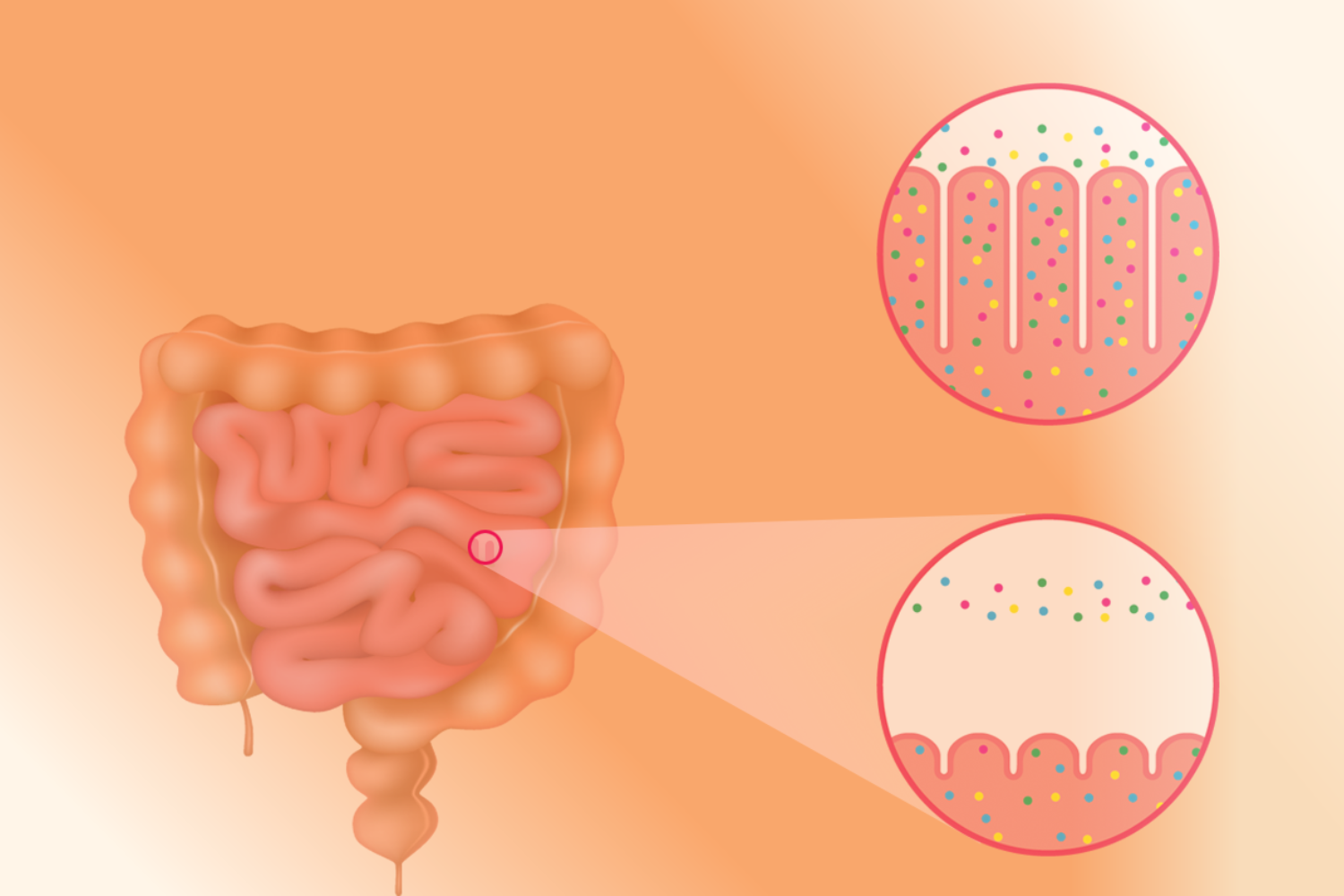
When a genetically susceptible individual ingests gluten, it is not fully digested because of its high proline content. As a result, substantial amounts of gluten peptides go undigested. Gluten peptides that have not been digested pass the intestinal epithelial barrier and enter the lamina propria. Lamina propria is that thin layer of connective tissue that forms the lining of the mucosa.
The intestinal epithelial barrier in the human body is physiologically impermeable to macromolecules like gliadin. Still then, why does it fail to prevent autoimmune reactions leading to the development of celiac disease? It is because; gliadin interacts with intestinal cells in patients with a hereditary predisposition to celiac disease, causing the inter-enterocyte tight junctions (TJ) to disassemble. T-lymphocytes in the lamina propria thus get activated by gliadin peptides that penetrate through the epithelial barrier.
The undigested gluten peptides come into contact with tissue transglutaminase and antigen-presenting cells expressing DQ2 or DQ8 genes in the lamina propria. Almost everyone with celiac disease carries the major histocompatibility complex (MHC) class II Human Leucocyte Antigen (HLA) haplotypes, namely DQ2 or DQ8 gene.
The undigested gluten peptides then bind to DQ2 or DQ8, and some of these peptides are then delivered to the CD4+ T-cell population. Activated CD4+ T-lymphocytes produce high pro-inflammatory cytokines that undergo clonal expansion to secrete anti-gliadin and anti-tissue-transglutaminase antibodies. This leads to tissue damage.
More scientific research is yet to be done to get an accurate and clear picture of the pathogenesis of Celiac disease.
Conclusion
Celiac disease is undeniably still a contentious and complicated human condition. Gluten causes enteropathy in celiac patients, resulting in deterioration of the mucosal surface and, as a result, improper food absorption. Presence of a wide range of clinical symptoms and the involvement of multiple human systems, celiac disease is sometimes referred to as a syndrome. Patients with Type 1 Diabetes, Down syndrome, Addison’s disease, psoriasis, and many other autoimmune diseases are at high risk of developing celiac disease. Diarrhea, fatigue, heartburn, gradual weight loss, bloating, constipation, and change in the color of teeth are some of the symptoms out of many symptoms of celiac disease. The celiac disease currently has no specific drug or treatment. The only solution is to take the necessary precautions. Gluten-containing foods such as beer, bread, cereals, pancakes, sauces, and many others should be avoided by celiac patients. Before eating, people should carefully read food labels. Scientists are carrying out research and studies to improve the quality of life of celiac disease patients.
References
- Van Heel, D. A., & West, J. (2006). Recent advances in coeliac disease. Gut, 55(7), 1037–1046. https://doi.org/10.1136/gut.2005.075119
- Parzanese, I., Qehajaj, D., Patrinicola, F., Aralica, M., Chiriva-Internati, M., Stifter, S., Elli, L., & Grizzi, F. (2017). Celiac disease: From pathophysiology to treatment. World journal of gastrointestinal pathophysiology, 8(2), 27–38. https://doi.org/10.4291/wjgp.v8.i2.27
- Gujral, N., Freeman, H. J., & Thomson, A. B. (2012). Celiac disease: prevalence, diagnosis, pathogenesis and treatment. World journal of gastroenterology, 18(42), 6036–6059. https://doi.org/10.3748/wjg.v18.i42.6036
- Kagnoff MF. Overview and pathogenesis of celiac disease. Gastroenterology. 2005 Apr;128(4 Suppl 1):S10-8. doi: 10.1053/j.gastro.2005.02.008. PMID: 15825116.
- Villanacci, V., Not, T., Sblattero, D., Gaiotto, T., Chirdo, F., Galletti, A., & Bassotti, G. (2009). Mucosal tissue transglutaminase expression in celiac disease. Journal of cellular and molecular medicine, 13(2), 334–340. https://doi.org/10.1111/j.1582-4934.2008.00325.x